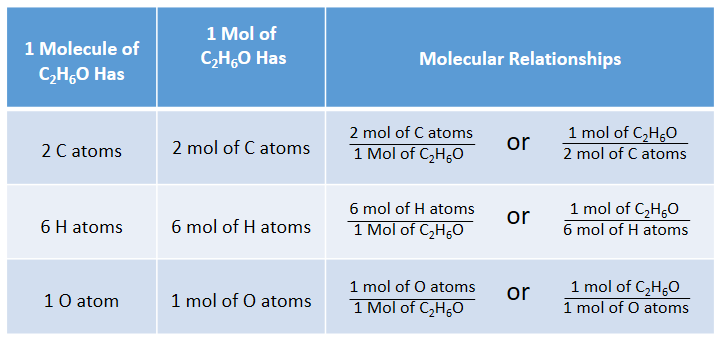
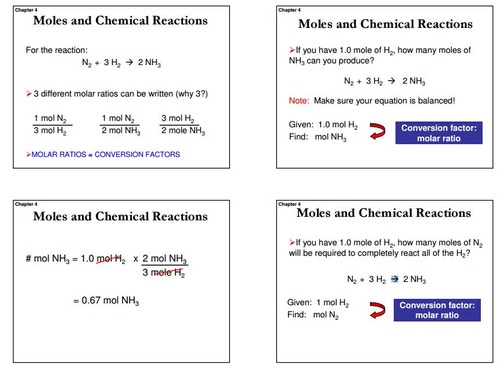
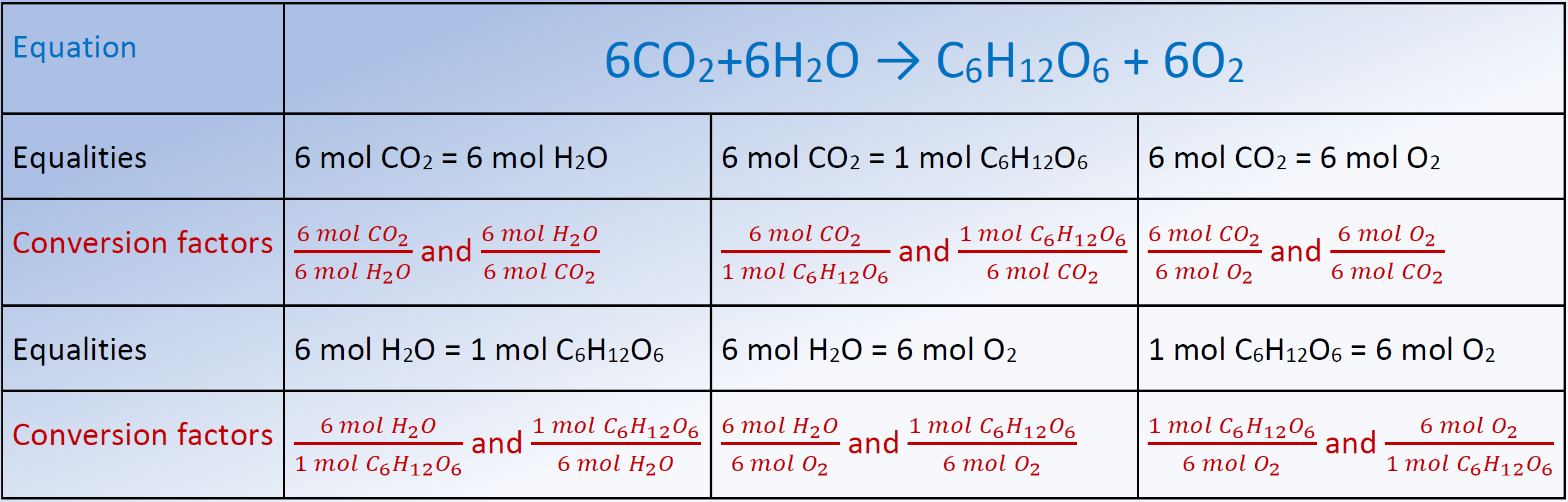
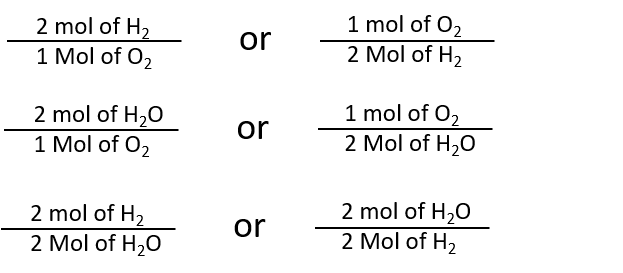
Question Video: Calculating the Percentage by Mass of Water in Alum Given Its Chemical Formula | Nagwa

Question Video: Identifying a Precipitating Agent for the Gravimetric Analysis of Chloride Ions | Nagwa

How to Find Moles of Product from Moles of Reactant using a Chemical Equation | Chemistry | Study.com

Convert 22 g of carbon dioxide `(CO_(2))` into moles. (Atomic masses : `C = 12 u, O = 16 u`) - YouTube
2 mol of Hg(g) is combusted in a fixed volume bomb calorimeter with excess of O2 at 298 K - Sarthaks eConnect | Largest Online Education Community
2 moles of an ideal gas at temp 27 degree c is heated isoermall from volume v to 4v .if R=2 cal/mol then the heat input in the process is approximately