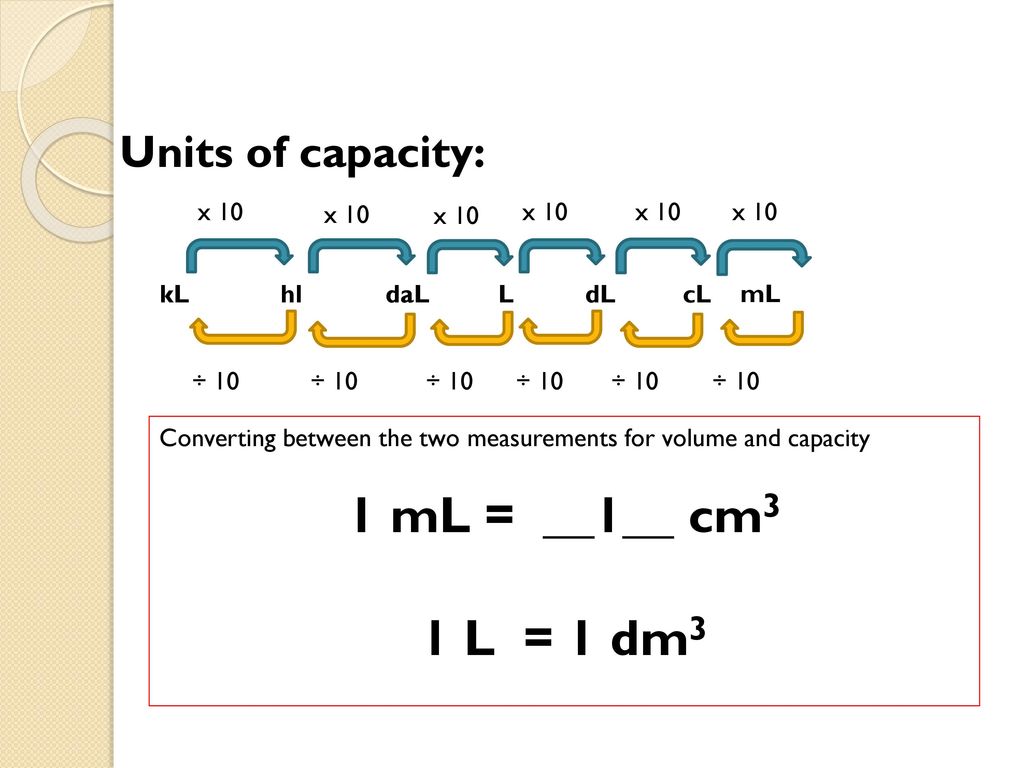
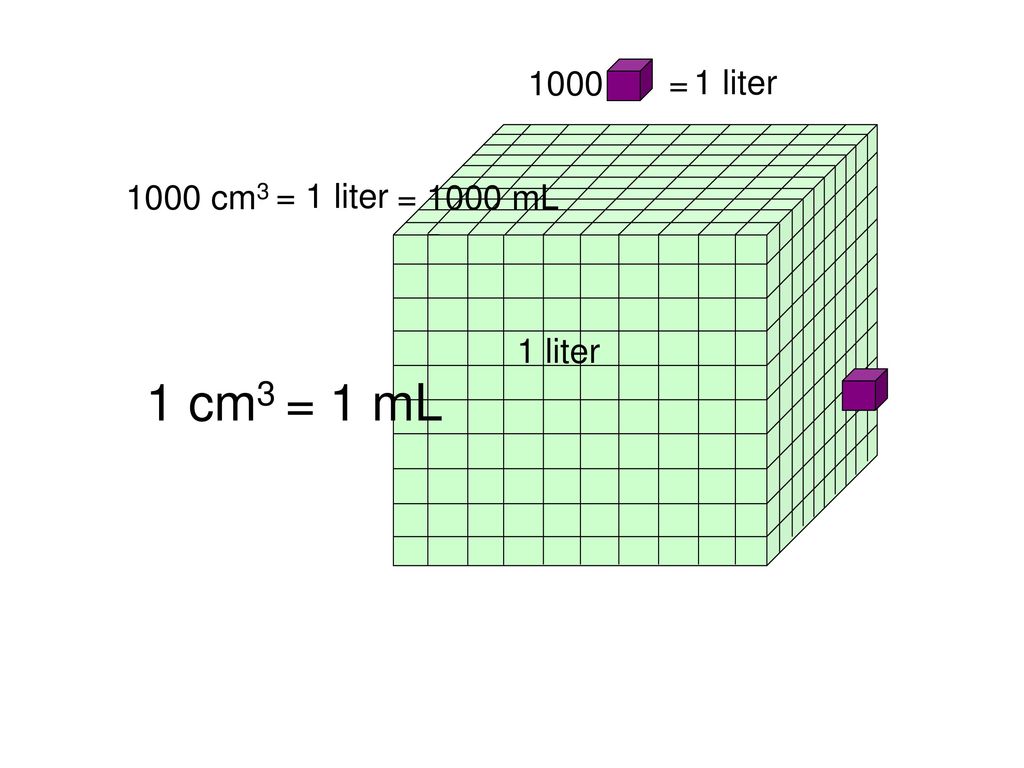
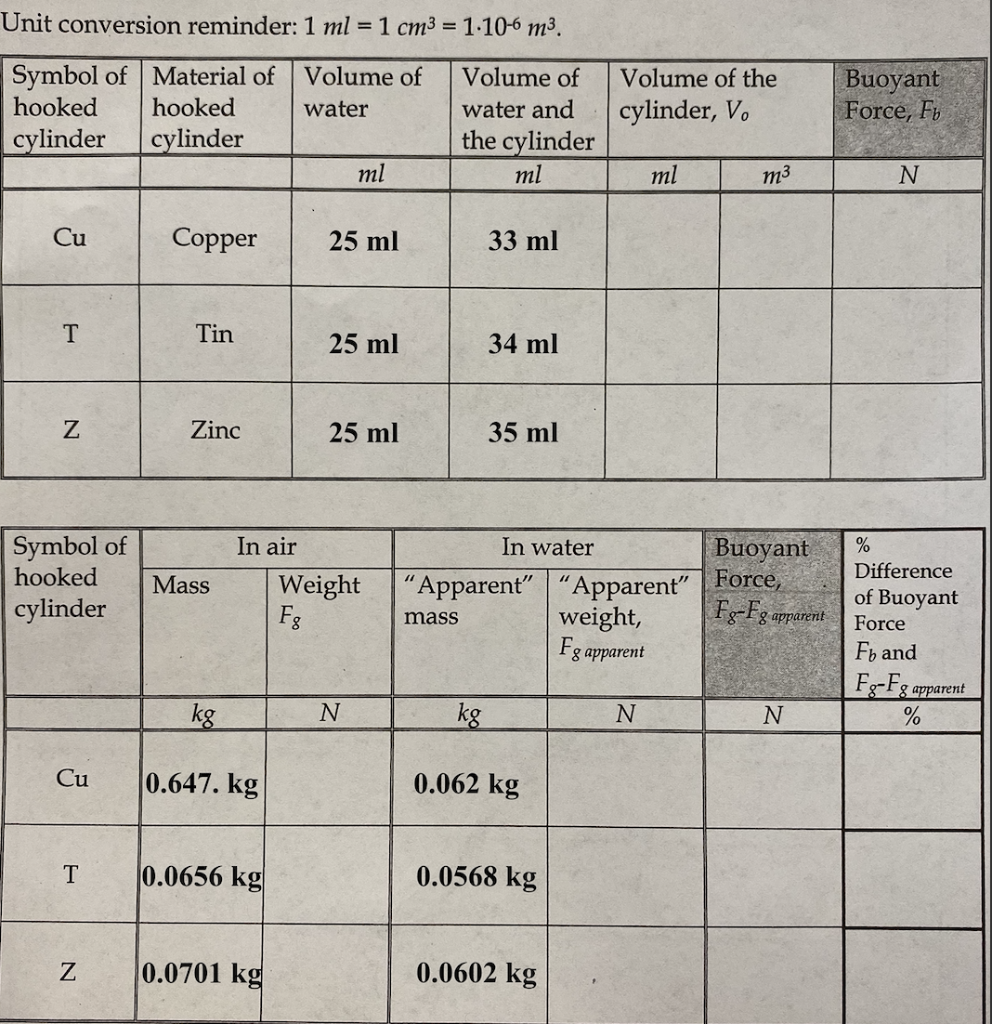
SOLVED: Determine the density (g/mL) for each of the following: A 14.3-cm3 sample of tin has a mass of 0.104 kg.Express your answer with the appropriate units.
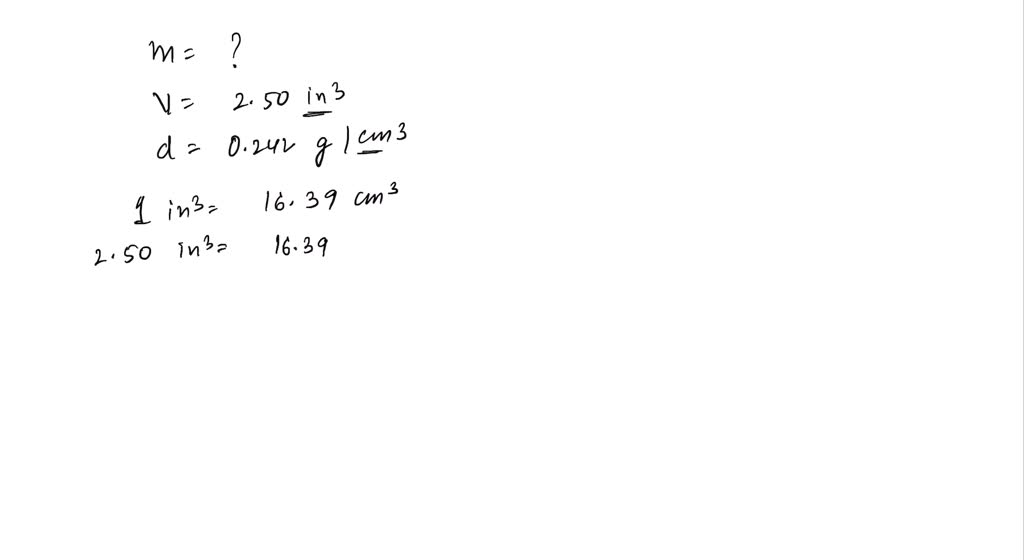
SOLVED: The mass unit associated with density is usually grams. If the volume (in mL or cm3) is multiplied by the density (g/mL or g/cm3) the volume units will cancel out, leaving