
EiKO BBA Inside Frosted Light Bulb (Pack of 2), 8500 Lumens, 120 Voltage Rating, 250 Watts, 2.08 Amps, Medium Screw (E26) Base, A-21 Bulb Type, C-9 Filament, 4.77"/121.2mm MOL - - Amazon.com

Quantum Mechanics/Molecular Mechanics (QM/MM) Calculations Support a Concerted Reaction Mechanism for the Zika Virus NS2B/NS3 Serine Protease with Its Substrate | The Journal of Physical Chemistry B

SOLVED: 'Question 6 of 6 Submit If 30.0 g of Nal (MM = 149.89 g/ mol) are added to a 500.0 mL volumetric flask, and water is added to fill the flask;
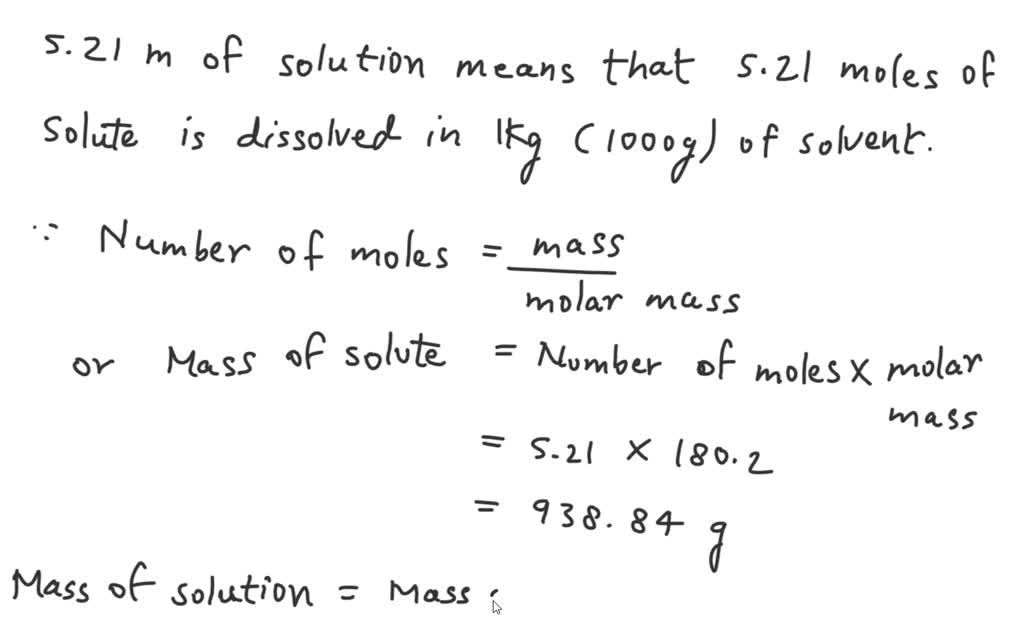
SOLVED: An aqueous solution of glucose (MM = 180.2 g/mol) has a molality of 5.21 m and a density of 1.20 g/mL. What is the molarity of glucose in the solution?

Machine Learning in QM/MM Molecular Dynamics Simulations of Condensed-Phase Systems | Journal of Chemical Theory and Computation

SOLVED: What is the concentration in molarity of a solution which is 1.53 %m/v acetone (MM = 58.08 g/mol ) in ethanol (MM = 46.07 g/mol)?

SJLITE ECO SAC T8 MIRROR OPTIC LOUVRE FITTING 220 SERIES (SURFACE) (METRIC) ECO SAC 2A 220/S MM MOL SJLITE Selangor, Malaysia, Kuala Lumpur (KL), Seri Kembangan Supplier, Suppliers, Supply, Supplies | E S L











